CMRMP scans and monitors for evolving state, Federal, and international regulatory drivers that may limit access to critical chemicals and materials. Below is an overview of select regulations that CMRMP actively tracks.
U.S. Environmental Protection Agency (EPA) Toxic Substances Control Act (TSCA)
Section 6 of TSCA provides EPA with the authority to prohibit or limit the manufacture, processing, distribution in commerce, use, or disposal of a chemical that presents an unreasonable risk to human health or the environment. CMRMP leads Department engagement with EPA to weigh in on the validity of the underlying science early in the rulemaking process and inform EPA of mission critical uses. The process shown below provides an overview of opportunities to engage within the TSCA process.
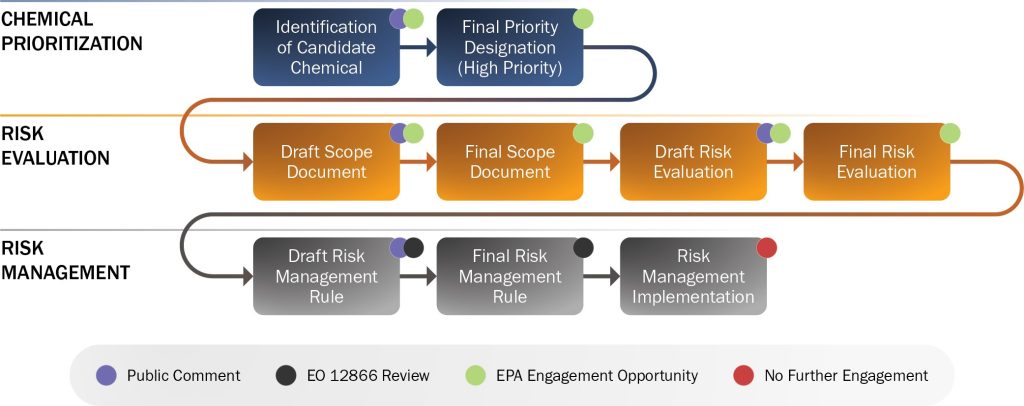
European Union (EU) Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH) Regulation
The European Chemicals Agency (ECHA) implements the EU REACH
regulation. The regulation places the burden of proof on companies to demonstrate how their substances – that are manufactured and marketed in the EU – can be safely used and to manage any risks associated with them. If risks cannot be managed, ECHA can restrict or ban the import, manufacturing, and use of hazardous substances.
Request Information
For questions or to share concerns on emerging chemical issues, please contact us through the Contact POC page or email our program mailbox: osd.pentagon.ousd-atl.mbx.cmrmp@mail.mil

EPA Resources
Risk Evaluation Process for Existing Chemicals under TSCA
Ongoing and Completed Risk Evaluations under TSCA
Chemicals undergoing Prioritization
ECHA Resources
Public Chemical Database under ECHA
Candidate List of Substances of Very High Concern
